SemaCyte® Microcarrier Workflow
SemaCyte® microcarriers turn adherent cells into optically barcoded, assay ready reagents that can be pooled and dispensed into standard microplates. The workflow is compatible with a wide range of adherent cell types, including engineered cell lines and more complex models, and supports both manual assays and automated 384 well screening. SemaCytes are fully compatible with common transfection, fixation, staining, and high content imaging protocols, with readouts that plug into standard analysis pipelines.
SemaCytes can be used in plate reader assays to leverage assay ready, miniaturised adherent cells. Note that cell multiplexing and barcode decoding require brightfield imaging, so they are not supported in plate reader workflows.
Seed & Grow
Cells are seeded onto immobilised SemaCyte® arrays in Seeding Dishes using standard culture practice, then incubated until the desired morphology and confluency are reached. Each SD20 Seeding Dish contains an array of ~50,000 immobilised SemaCytes with one optical barcode.

Release, Purify & Freeze
SemaCytes with cells are released from the dish by gentle agitation, collected into a tube, and purified with the SemaPure15 to remove debris and unattached cells.
Purified SemaCytes can be aliquoted and cryopreserved as batches of adhered, assay ready cells for on demand use. Cells are assay ready about 1 hour after thawing.

Pool, Dispense & Assay
Fresh or frozen SemaCytes with different barcodes and cells are pooled and dispensed into 96 well or 384 well plates using standard liquid handling. The SemaPlate orients SemaCytes and pulls them to the bottom of the well. Once positioned, downstream assay steps can be run without the magnet. Recommended densities are typically 10 to 200 SemaCytes per well in 384 well plates, depending on the assay.

Automation: SemaCytes are designed to plug into existing automation stacks. We have validated workflows for dispensing, washing, and staining using common tools such as the Multidrop, BioTek plate washers, FireFly, BRAVO, and ViaFlow. Contact us for detailed, instrument-specific protocols and recommended settings.
Image Analysis & Barcode Deconvolution
SemaCytes carry optical barcodes in the microcarrier rim, using a QR-code like pattern that is visible in the brightfield channel. After imaging, software first detects and isolates individual microcarriers within each well image, then decodes the barcode by matching it to a reference to assign cell identity.
Barcode deconvolution is supported through Semarion’s Semalyse software and through microscopy software workflows, including Harmony and Signals Image Artist, enabling cell-type resolved datasets from multiplexed wells.
Following deconvolution, analysis can be performed directly on cells within each SemaCyte, or the workflow can export tiled, barcode-separated image sets for downstream processing in any preferred software, including ImageJ and CellProfiler.
Use Cases Across Screening and Profiling
SemaCyte® microcarriers enable a wide range of adherent cell assays for target validation, screening, and profiling in standard microplates. The platform has been validated across diverse formats and readouts, including functional and mechanistic assays, viability and proliferation assays, and phenotypic high-content imaging, with workflows that integrate with common automation and analysis pipelines.
High-Fidelity Antibody Selectivity Screen
3-plex antibody screening pools CHO cells expressing different target variants, orthologues, or controls to quantify binding specificity and selectivity.
This approach accelerates selectivity screening while reducing assay variability and dependence on cytometry.

Multi-Model Cell Painting
4-plex cell painting pools A549, HEK293, U2OS, and HeLa cells to profile distinct phenotypic responses in a single well.
This approach enables robust cell-type resolved profiling from 100–200 cells per line while reducing plates, reagents, and imaging time.

Robust DNA Damage Response Profiling
4-plex DDR assay pools A549, U87MG, U2OS, and control cells to quantify γH2AX nuclear intensity and generate dose response curves and EC50 values.
This approach delivers reproducible DDR profiles from ~80 cells per condition while improving throughput and assay consistency.

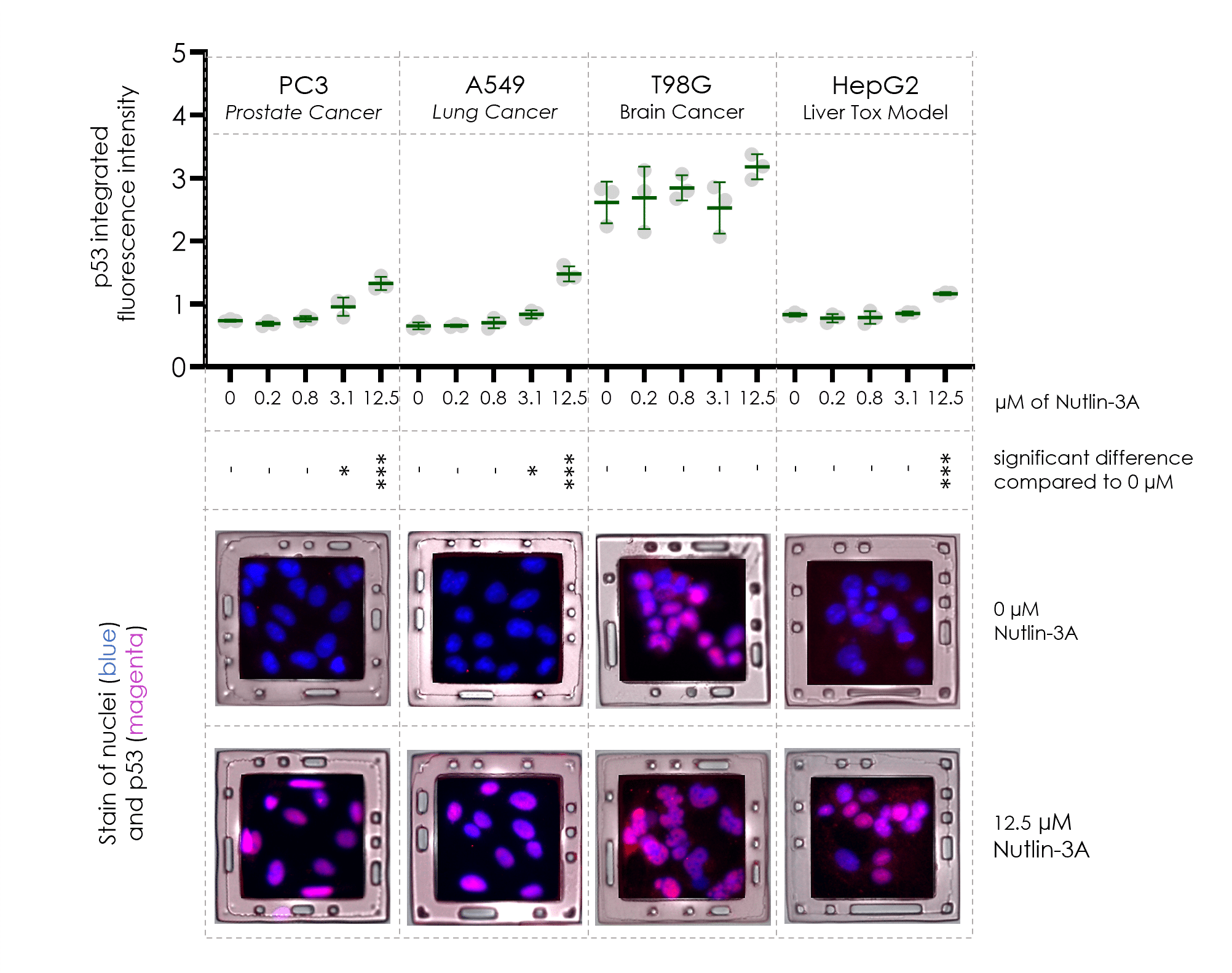
Faster Cell Panel Screens
4-plex mechanistic assay pools A549, PC3, T98G, and HepG2 cells to evaluate differential p53 induction by Nutlin-3a.
This approach reduces assay time by 2-fold and reagent cost by 4-fold.
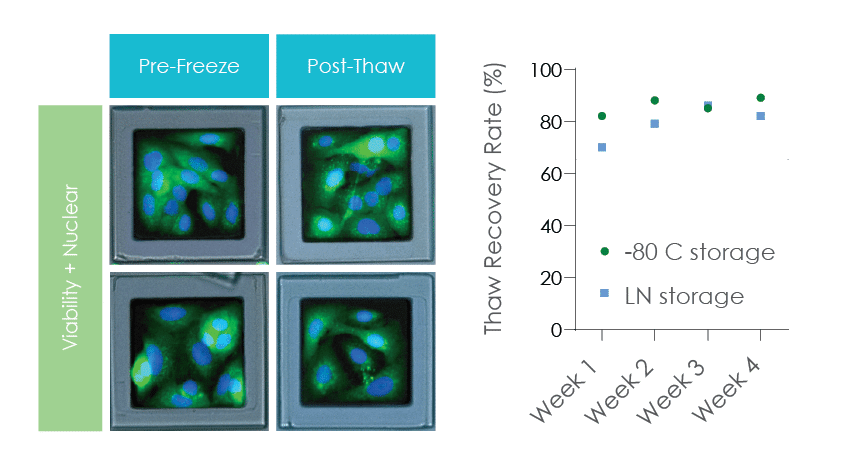
High Recovery for Adherent Assay-Ready Cells on SemaCytes
A549 lung cancer cells attached to SemaCyte microcarriers for 24 hours and batch frozen in cryovials showed consistant >75% recovery rates.
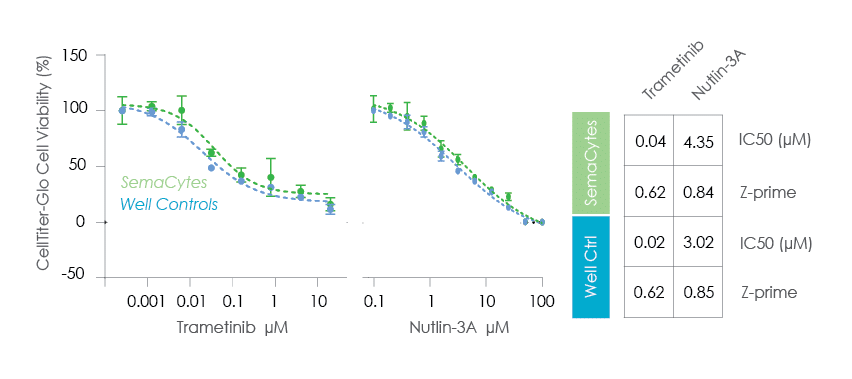
Rapid Viability & Proliferation Assays
Assay-ready cells on SemaCytes were used for 24h viability or 72h proliferation assays.
This approach removes the 24h cell seeding and attachement step.
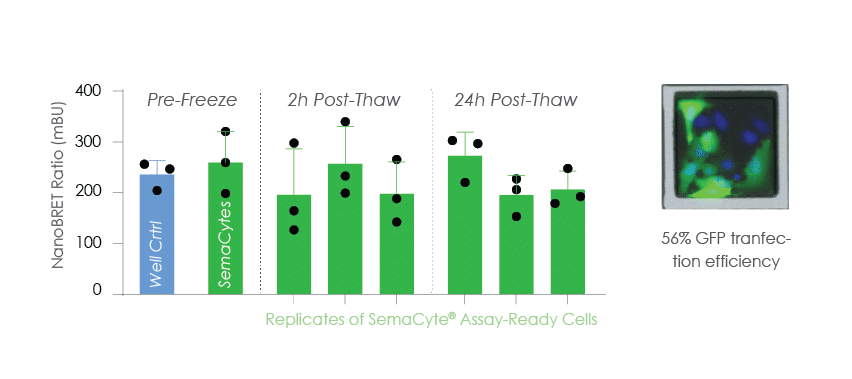
Rapid Mechanistic Assay
NanoBRET PPI cells were batch prepared on SemaCytes and frozen for on demand use in mechanistic assays.
This approach removes 3 days transfection preparation time for ultra-fast drug data generation.
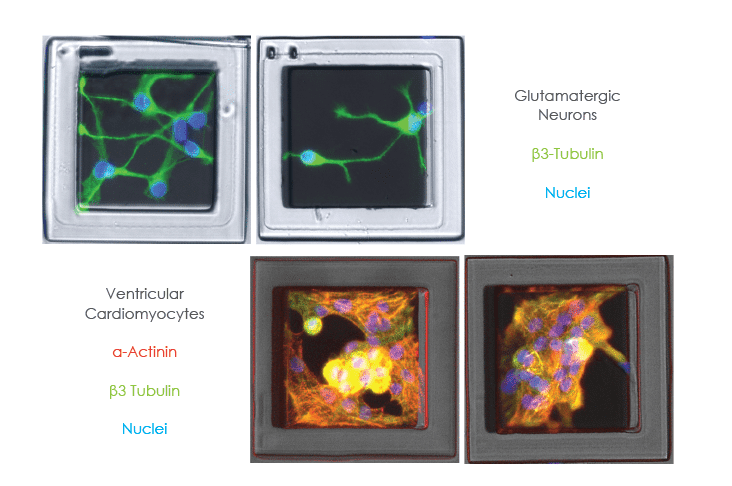
Miniaturised Complex Assays
iPS or patient derived cells can be grown on SemaCytes and dispensed at a low density in 96 or 284-well plates.
This approach can reduce the amount of cells-per-well by 5- to 50-fold to increase the number of experiments per cell sample.